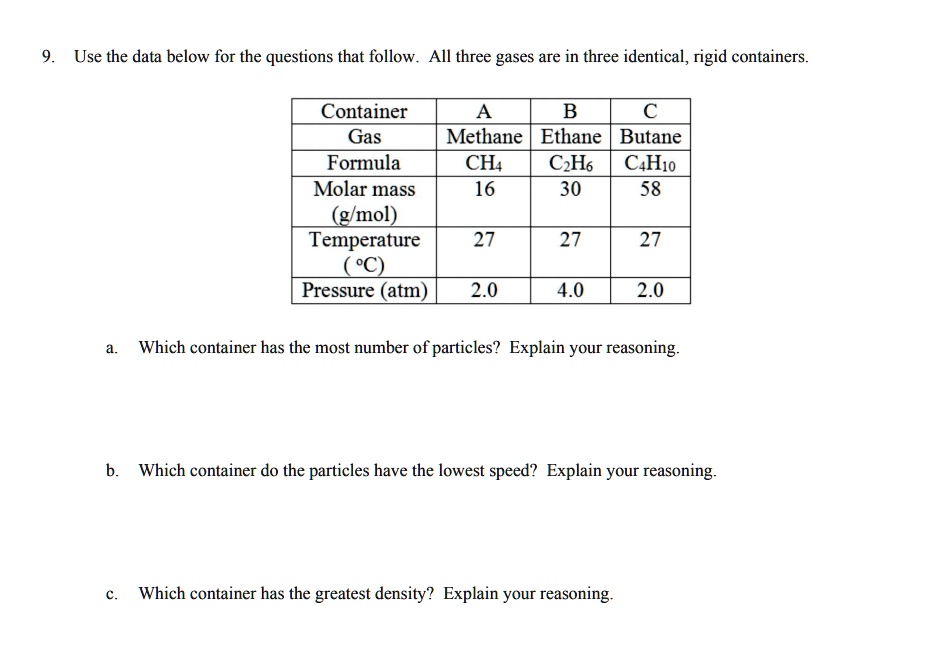
SOLVED: Use the data below for the questions that follow All three gases are in three identical, rigid containers Container Gas Methane Ethane Butane Formula CH C,Ho C4H1o Molar mass 16 30

SOLVED: Butane (C4H10) is the liquid fuel in lighters. How many grams of carbon are present within a lighter containing 7.25 mL of butane? (The density of liquid butane is 0.601 g/mL.)

a) Bulk modulus and (b) density of common reservoir gases methane (C 1... | Download Scientific Diagram
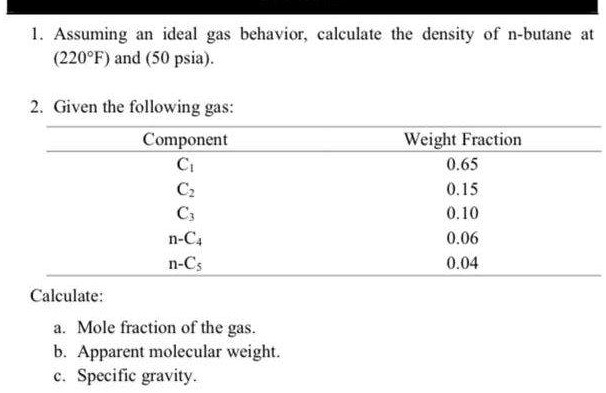







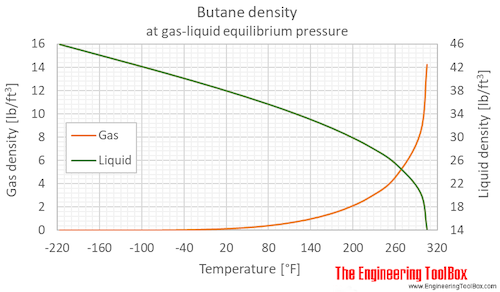



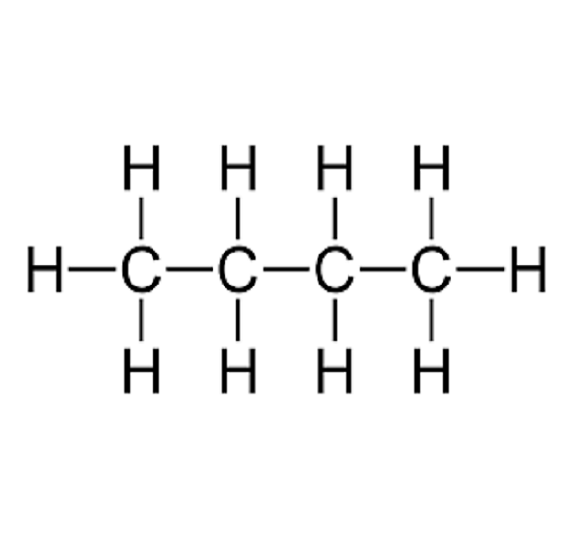

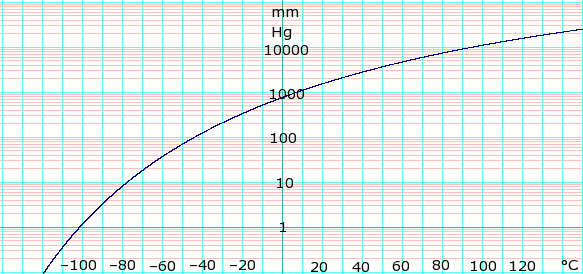
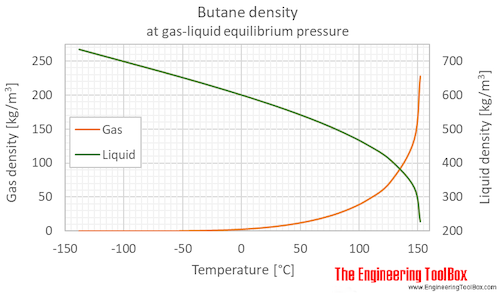
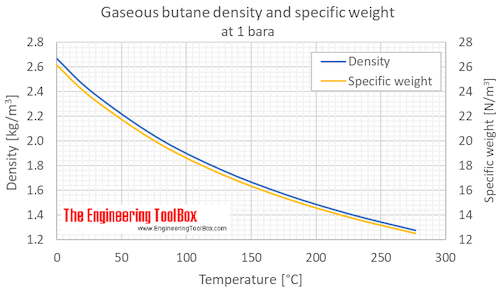

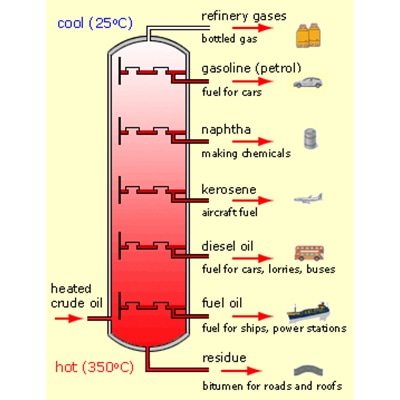

![Solved (References] Butane, C4H10, is an easily liquefied | Chegg.com Solved (References] Butane, C4H10, is an easily liquefied | Chegg.com](https://media.cheggcdn.com/study/da1/da115387-e0c5-4ae9-9f37-efe83e35bbdb/image.png)
